CURRENT ISSUE
June, 2026
No. 111 (6)
2024 Impact Factor: 7.9
2024 Journal Citation Indicator: 1.9
2024 CiteScore: 11.3
2024 Journal Citation Indicator: 1.9
2024 CiteScore: 11.3
EDITOR'S PICKS
Perspective Article
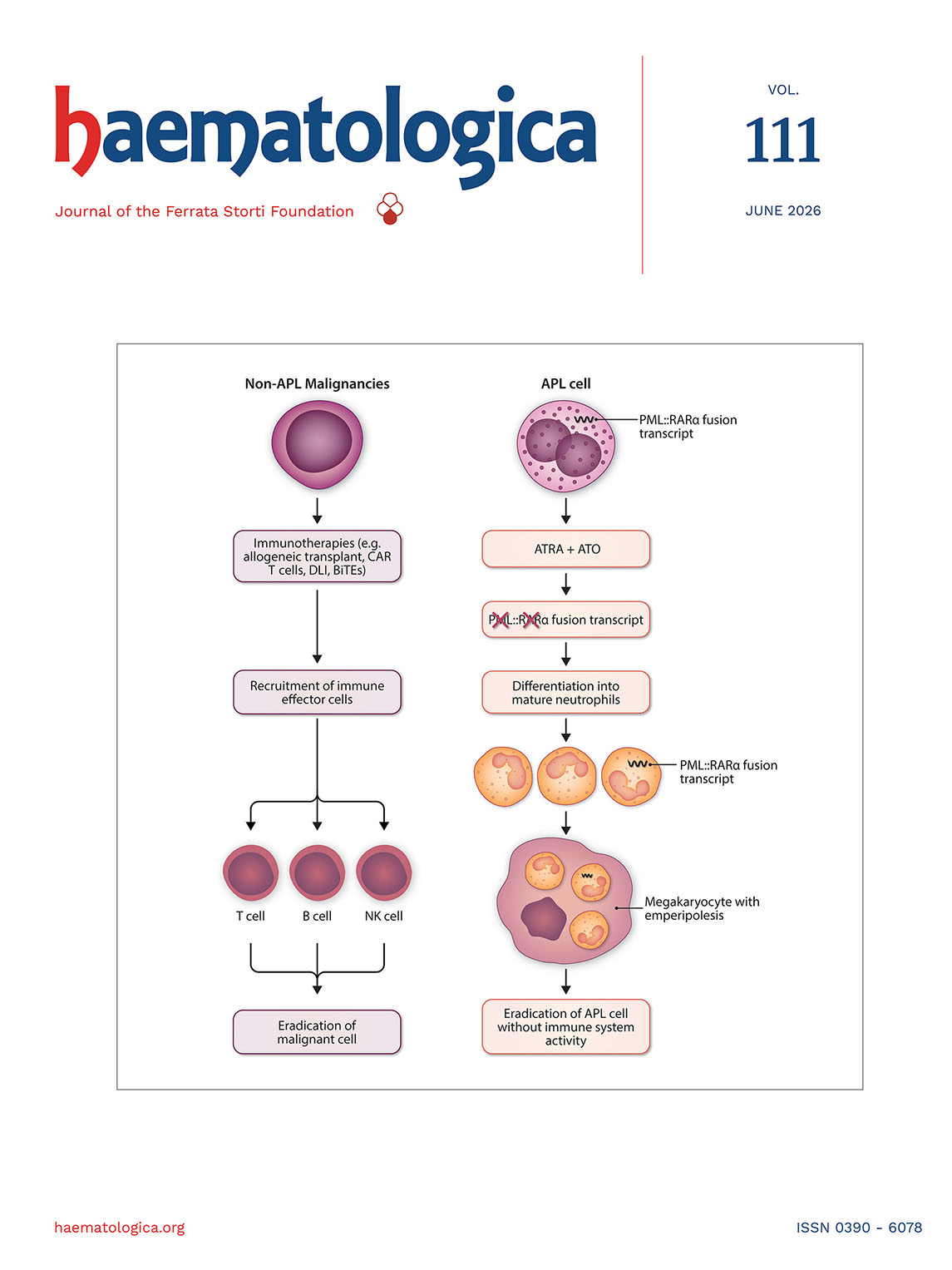
Optimal management of elderly/old Ph+ acute lymphoblastic leukemia patients
ARTICLES IN THREE SENTENCES
Article
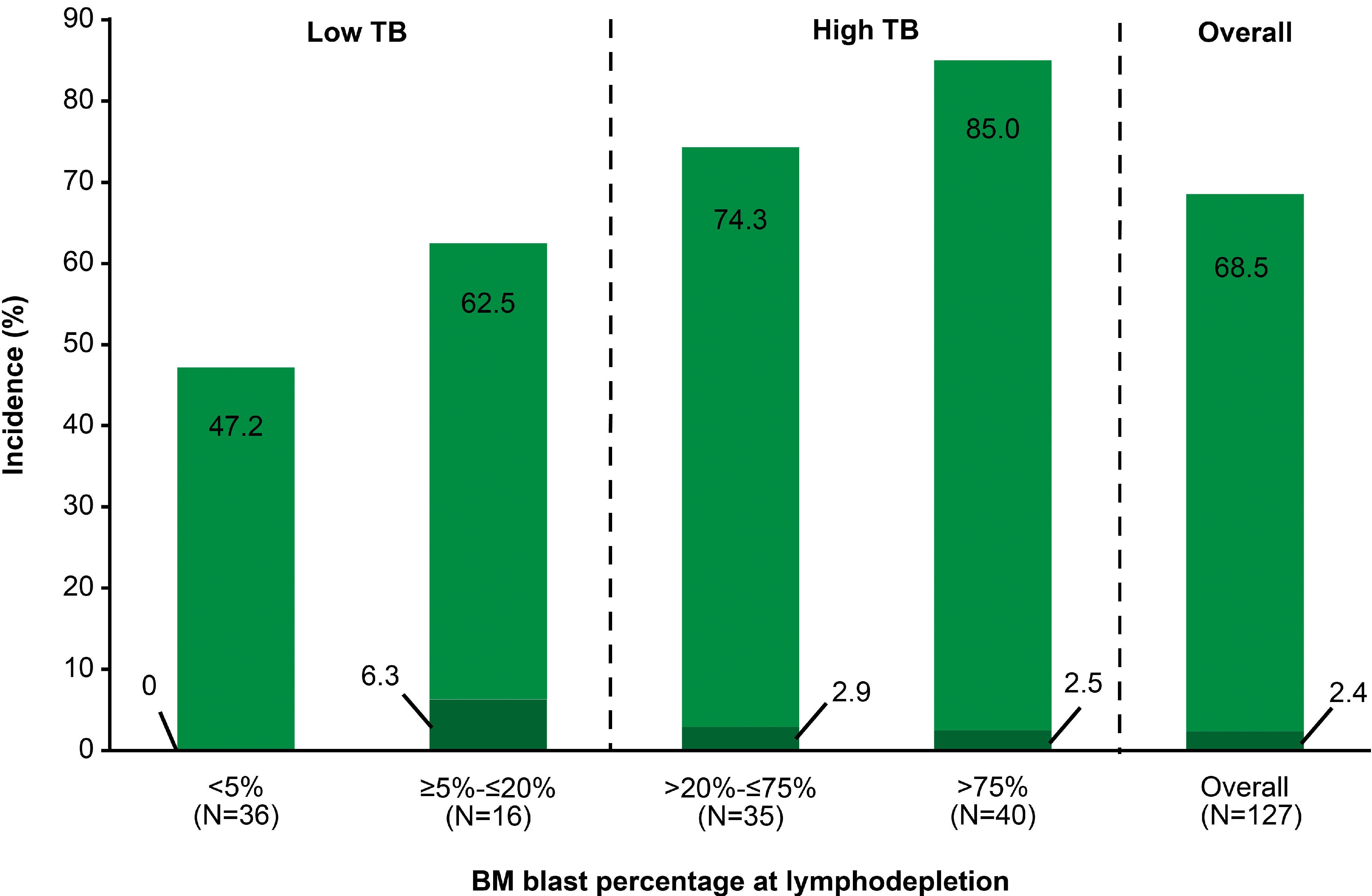
Tumor burden-guided dosing contributes to mitigation of immunotoxicities following treatment with obecabtagene autoleucel in adult patients with relapsed/refractory B-cell acute lymphoblastic leukemia
Obecabtagene autoleucel (obe-cel) is a CD19-directed chimeric antigen receptor (CAR) T-cell therapy for relapsed or refractory B-cell acute lymphoblastic leukemia (B-ALL). In this study, the authors evaluated a tumor burden-guided dosing strategy for obe-cel in adult patients with relapsed/refractory B-ALL. Results show that adjusting CAR T-cell dosing reduces cytokine release syndrome and neurotoxicity while maintaining antileukemic activity.
Article
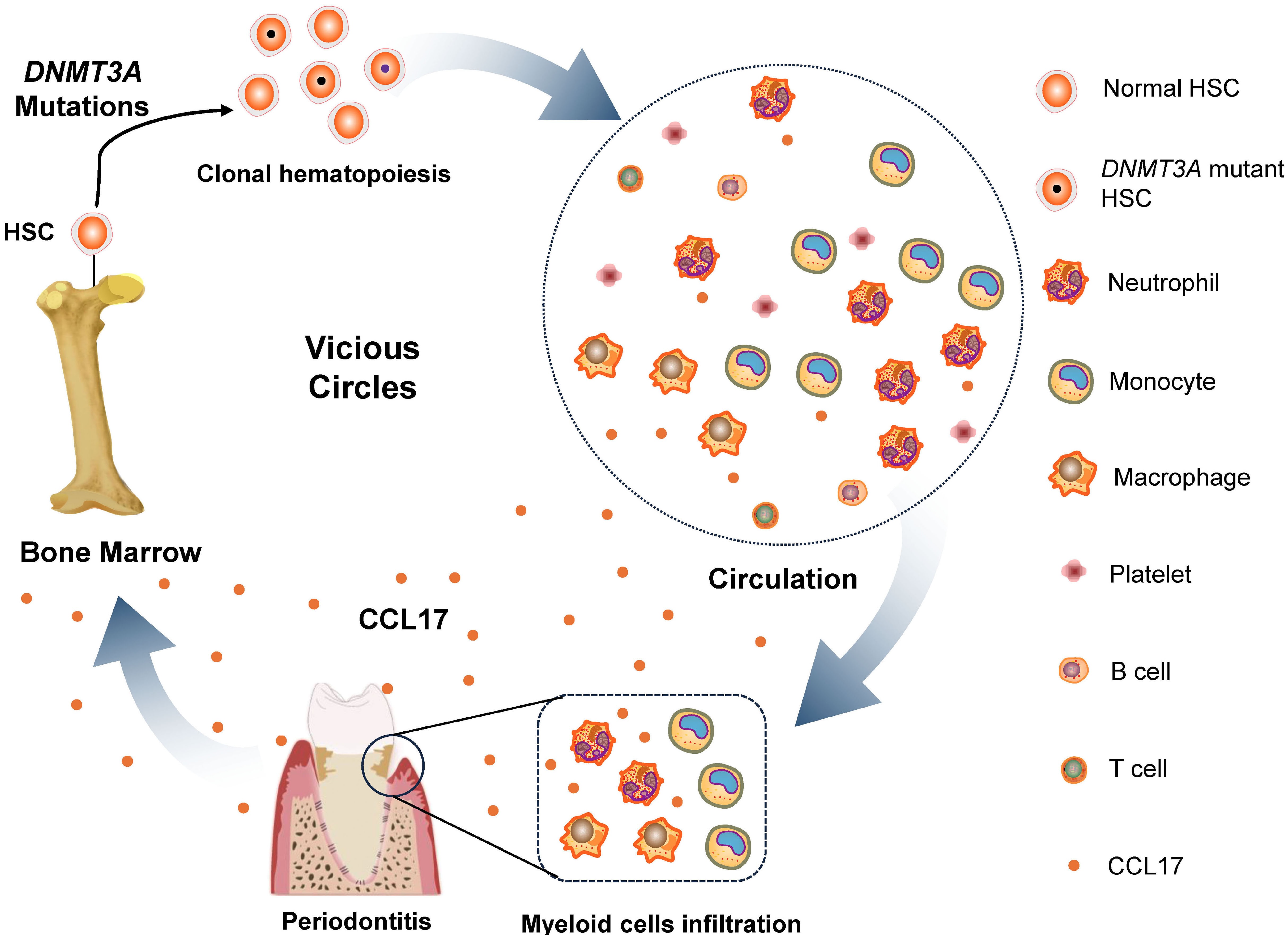
Ligature-induced periodontitis promotes Dnmt3aR878H-driven clonal hematopoiesis
Clonal hematopoiesis (CH) driven by DNMT3A mutations is influenced not only by intrinsic genetic alterations but also by inflammatory signals. In this study, the authors used a ligature-induced periodontitis model to investigate whether chronic oral inflammation promotes expansion of Dnmt3aR878H-mutant hematopoietic clones. They found that periodontitis accelerates Dnmt3a-mutant CH through inflammation-associated mechanisms.
Letter
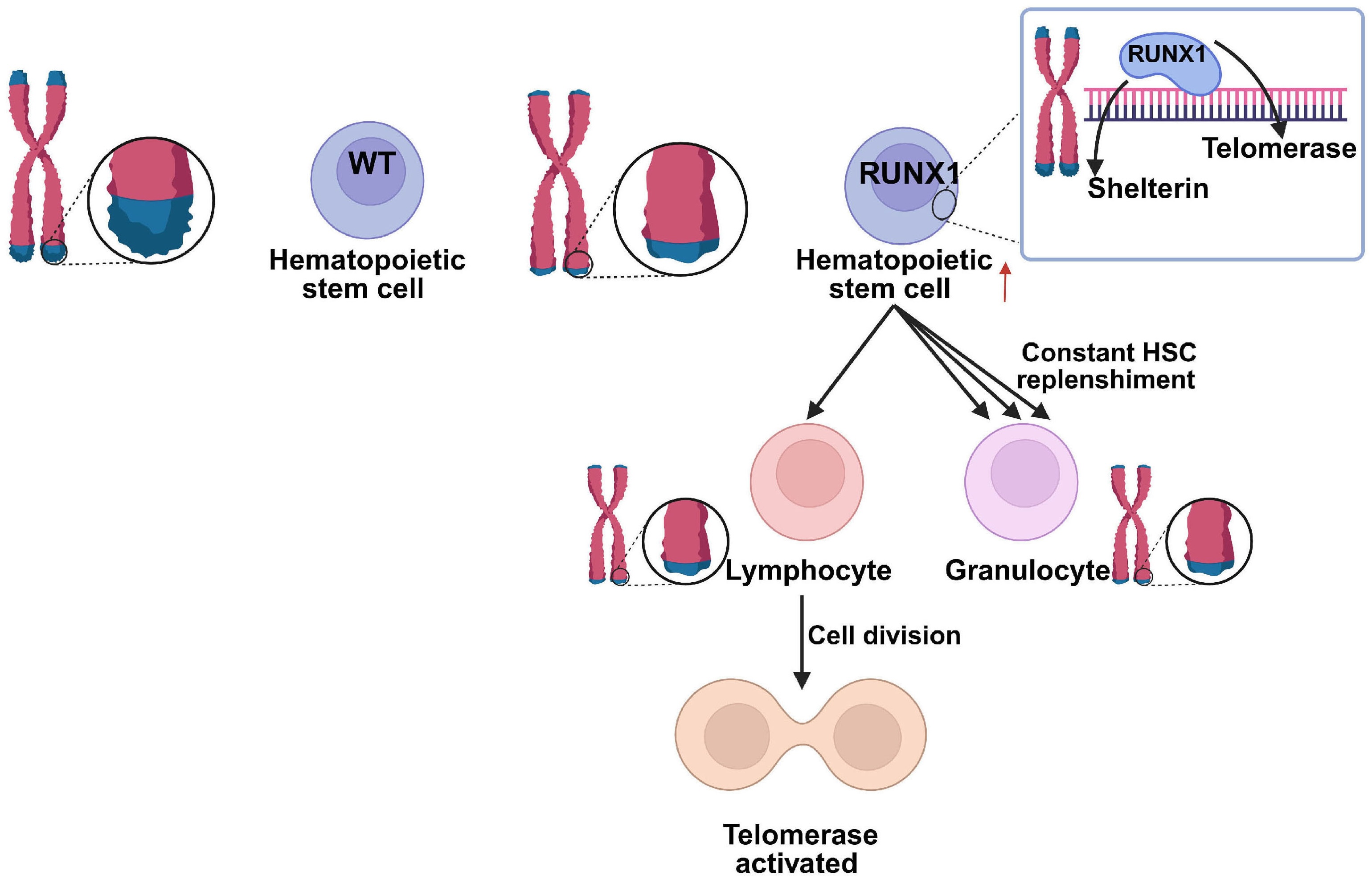
Telomere attrition is common in patients with germline RUNX1 pathogenic variants
Germline pathogenic variants in RUNX1 predispose to inherited platelet disorders and an increased risk of hematologic malignancies, although the mechanisms underlying disease progression remain incompletely understood. In this study, the authors investigated telomere length in patients carrying germline RUNX1 variants. They found that telomere attrition is frequent in RUNX1-mutated individuals, suggesting that impaired telomere maintenance may represent an additional mechanism promoting hematopoietic instability and leukemic predisposition.
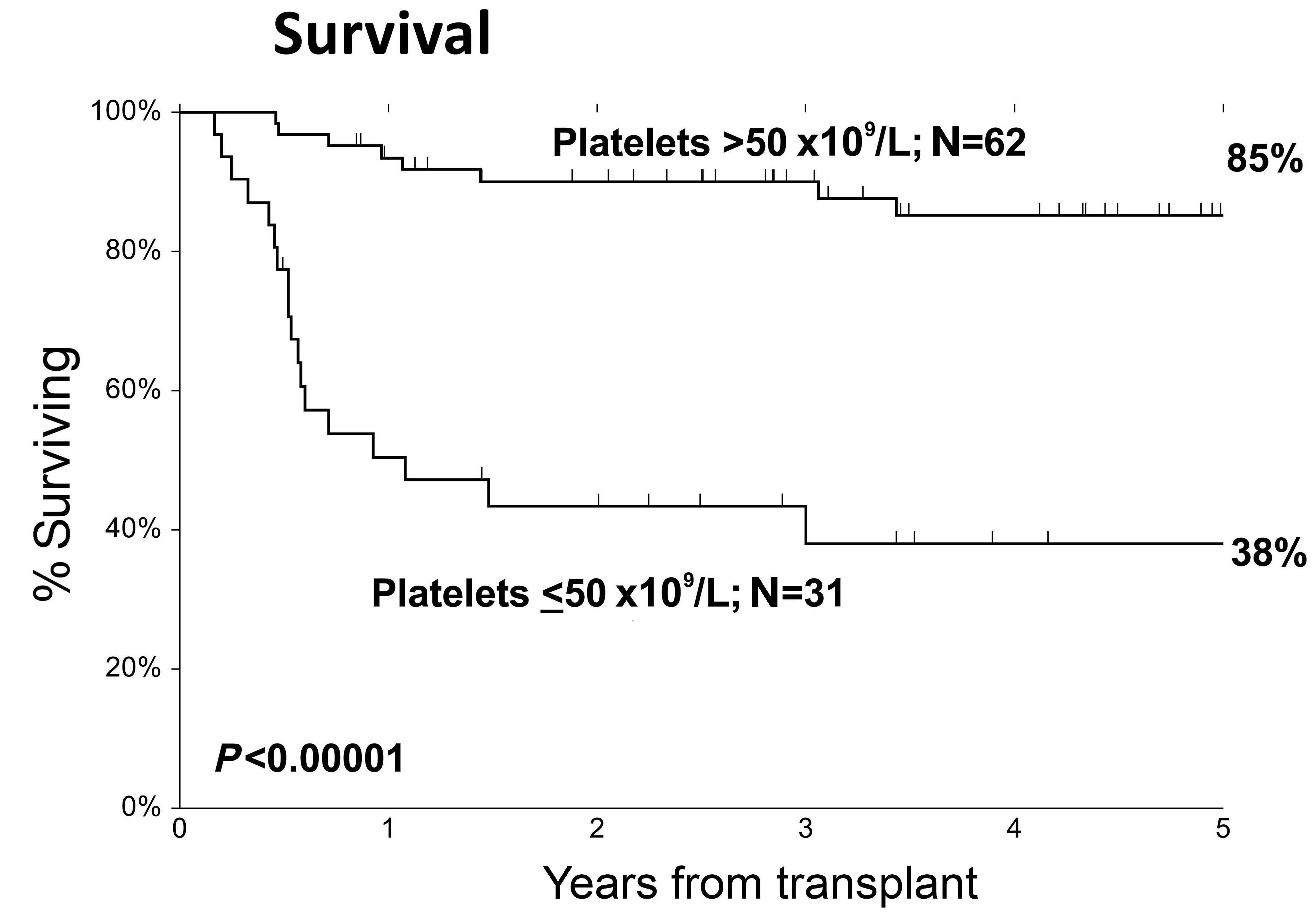
Article
Platelet recovery delay and survival in patients with myelofibrosis undergoing allogeneic hemopoietic stem cell transplantation
Allogeneic hematopoietic stem cell transplantation remains the only potentially curative treatment for myelofibrosis, although post-transplant hematologic recovery strongly influences patients’ outcomes. In this study, the authors evaluated the association between platelet recovery kinetics and survival. They found that delayed platelet recovery was associated with inferior survival and poorer transplant outcomes.
TAKE ADVANTAGE FROM HAEMATOLOGICA