Abstract
Introduction. Nowadays continuous treatment until disease progression or unacceptable toxicity remains the main approach in multiple myeloma (MM). However, prolonged exposure may lead to cumulative toxicity, treatment fatigue and quality-of-life impairment. Indeed, in clinical practice, a not negligible number of patients discontinues therapy due to medical, personal, or logistic reasons. Recent evidence from time-limited regimens has renewed interest in whether discontinuation may be feasible and safe in selected cases.
Methods. The ongoing, multicenter ODISSEY study (Observational stuDY of multiple myeloma patientS who diScontinuEd therapY in clinical practice) was designed to describe clinical features and outcome of responding MM patients who discontinued treatment for reasons other than disease progression or death and were alive without progression for at least 12 months post-discontinuation.
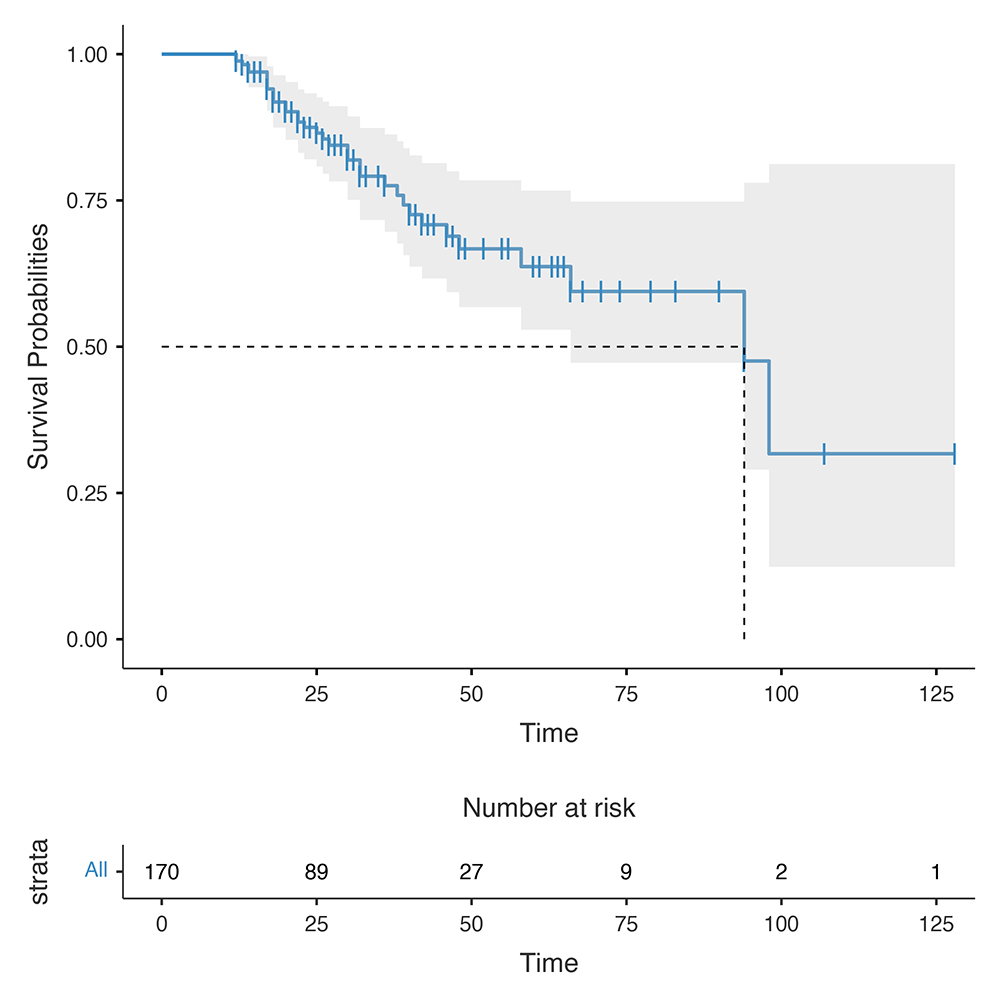
Results. One hundred and seventy patients (55.9% male) diagnosed between 2002 and 2024 have been so far enrolled. The median age at discontinuation was 69 years (range, 40-89). At diagnosis, 71 patients (47.3 %) were in ISS stage I, 38 (22.3%) in stage II, 16 (9.4%), in stage III; ISS was not available in 16 patients. R2-ISS was reported in 70 patients (I, 31 patients; II, 31 patients; III, 8 patients), while extramedullary disease occurred in 6 patients. Treatment was discontinued during first, second and third or subsequent lines in 92 (54.1%), 57 (33.6%) and 21 (12.3%) patients, respectively. Specifically, the most frequent first line treatments discontinued included lenalidomide or thalidomide maintenance after autologous stem cell transplantation (63%), while combinations of lenalidomide and dexamethasone with daratumumab (DRd) or carfilzomib (KRd) where those more frequently interrupted during second line therapy (57.9%). The median duration of treatment before discontinuation was 18 months (range, 1–85), overlapping the median treatment duration from best-response achievement to discontinuation. At suspension of therapy, 155 patients (91.2%) were in VGPR or better (16 sCR; 93 CR; 46 VGPR). Main reasons for discontinuation were shared medical decision (26.5%), newly acquired comorbidities (21.8%), non-hematologic toxicity (15.3%), patient’s choice (12.9%), hematologic toxicity (12.3%), and other causes (11.2%). At a median follow-up of 28 months (range, 12-162) from treatment discontinuation, relapse-free survival (RFS) was 99% (95% CI, 97%-100%), 78% (95% CI, 70%-86.3%) and 64% (95% CI, 53%-76.7%) at 12, 36 and 60 months respectively. Median RFS was 94 months (95% CI 66–NR) (Figure 1). Among 35 patients who relapsed (20.6%), 31 received a new treatment and VGPR or better was re-obtained in 80.8% of cases. To date, 157 (92.4%) patients are still alive, and only one death due to progression disease has occurred.
Conclusions. The ODISSEY study represents one of the first real-world experiences exploring treatment discontinuation in MM outside of clinical trials. Despite heterogeneous therapeutic settings, a significant subset of not heavily treated patients, with less advanced disease at diagnosis and good quality responses, achieved durable off-therapy remissions, thus suggesting that treatment cessation may not be detrimental, at least in selected cases. Further studies incorporating measurable residual disease assessment will be necessary to validate these preliminary findings and to identify the best possible target population.

Footnotes
Disclosures
No Conflict of interest.
Funding
No funding.
Article Information

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
