Abstract
Background. Systemic AL amyloidosis is a rare disorder characterized by deposition of misfolded monoclonal immunoglobulin light chains as insoluble amyloid fibrils in various tissues and organs, leading to life-threatening organ dysfunction. Treatment (tx) is mostly directed against plasma cells (PCs) and focuses on stopping production of amyloid-forming light chains. Daratumumab (Dara), a human IgGκ monoclonal antibody targeting CD38, combined with bortezomib, cyclophosphamide, and dexamethasone, is the only regimen indicated in the USA and Europe for the tx of newly diagnosed AL amyloidosis. Some pts become refractory to initial tx and most ultimately relapse. There is no currently approved tx regimen for pts with relapsed/refractory disease. Tec, a B-cell maturation antigen (BCMA) × CD3 bispecific antibody, has demonstrated deep and durable responses in multiple myeloma (MM). BCMA is expressed on MM and amyloid PCs; in vivo data confirm that soluble BCMA levels positively correlate with involved free light chain levels in pts with AL amyloidosis [Godara A, et al. Blood. 2019]. Moreover, results from 2 retrospective case series suggest that Tec may induce rapid and deep hematologic responses in heavily pretreated AL amyloidosis with no unexpected adverse events [Forgeard N, et al. Blood. 2024; Stalker M, et al. Blood. 2023], justifying prospective clinical trials with Tec in AL amyloidosis. The ongoing phase 2 TeclistAMY (EMN40) trial is evaluating efficacy and safety of Tec monotherapy in pts with previously treated AL amyloidosis.
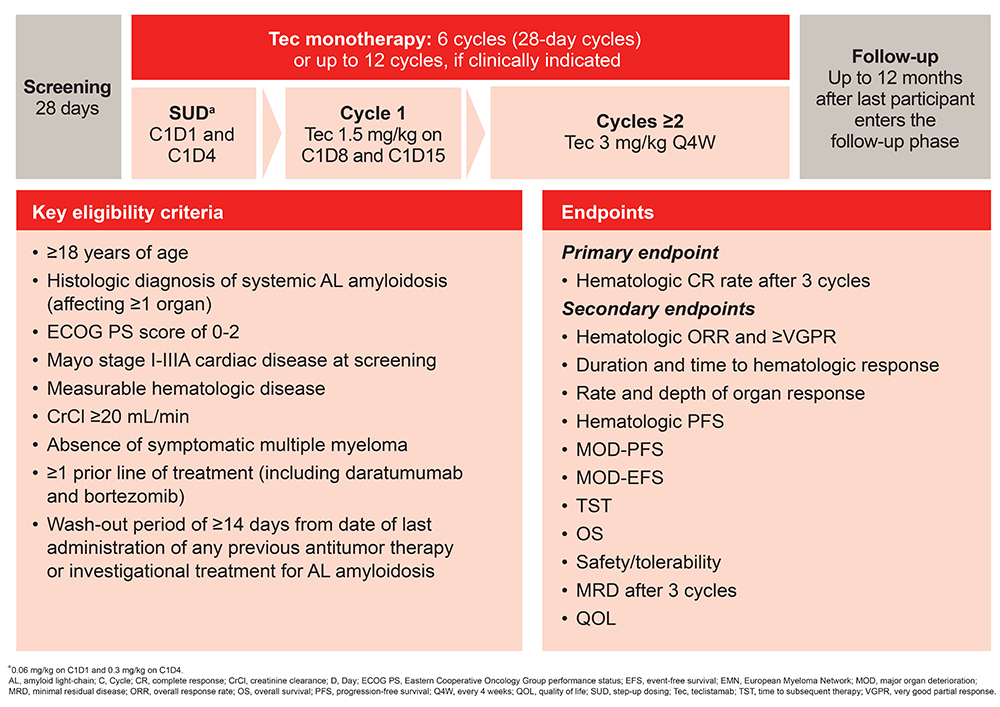
Methods. This ongoing open-label study aims to enroll 30 pts ≥18 yrs with histologic diagnosis of systemic AL amyloidosis, an ECOG performance status of 0-2, Mayo stage I-IIIA cardiac disease at screening, measurable hematologic disease, a creatinine clearance of ≥20 mL/min, and absence of symptomatic MM. Pts must have received ≥1 line of tx (including Dara and bortezomib), and a wash-out period of ≥14 days from date of last administration of any previous antitumor tx or investigational tx for AL amyloidosis. Following initial step-up dosing in Cycle (C) 1, Tec is administered at a dose of 1.5 mg/kg on Days 8 and 15 of C1; from C2 onwards, Tec is administered Q4W at 3 mg/kg (Figure). All pts undergo six 28-day cycles of tx. Pts may continue to receive tx for up to 12 cycles if clinically indicated and upon discussion between the investigator and sponsor. The primary endpoint is hematologic complete response rate after 3 cycles. Secondary endpoints include hematologic overall response rate, very good partial response or better, duration and time to hematologic response, rate and depth of organ response, hematologic progression-free survival (PFS), major organ deterioration–PFS, major organ deterioration–event-free survival, time to subsequent tx, overall survival, safety/tolerability, minimal residual disease, and quality of life.
Preliminary Results. The study is being conducted at 10 sites across 6 countries and is actively recruiting. The first pt was enrolled in July 2025. As of January 14, 2026, 8 of the 10 sites have been activated, with 22 patients screened, 18 enrolled, and 13 treated (ClinicalTrials.gov identifier: NCT06649695). A safety analysis was performed by an Independent Data Monitoring Committee after 6 pts completed ≥1 cycle of tx. No safety signals were observed, and the trial is continuing as planned. Updated results will be presented during the meeting.
Footnotes
Disclosures
No Conflict of interest.
Funding
No funding.
Article Information

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
