Abstract
Background. Daratumumab is increasingly used as a part of the induction therapy in patients with newly diagnosed multiple myeloma (NDMM) and is associated with deeper responses to treatment. Autologous stem cell transplantation (ASCT) remains a standard frontline procedure for transplant‑eligible patients. However, daratumumab, particularly in combination with lenalidomide, may negatively affect hematopoietic stem cell (HSC) mobilization. Pivotal trials evaluating daratumumab‑based induction regimens in NDMM have reported lower HSC yields in the experimental arms, leading to increased use of plerixafor to achieve successful mobilization and collection. Plerixafor is a selective, reversible antagonist of the CXCR4 receptor that inhibits its interaction with the ligand SDF‑1α, thereby promoting the release of HSCs into the peripheral blood and potentially overcoming the adverse effects of daratumumab on HSC mobilization. At our center, the target HSC dose for ASCT in NDMM patients is ≥2.5 × 10⁶ CD34⁺ cells/kg. Standard mobilization consists of granulocyte‑colony stimulating factor (G‑CSF) alone, administered subcutaneously at a dose of 10 µg/kg body weight daily on days 1–4. Peripheral blood (PB) CD34⁺ cell counts are assessed on day 4 after initiation of stimulation. A threshold of ≥20 × 10⁶ CD34⁺ cells/L in PB is required to proceed to apheresis. Mobilization failure is defined as failure to achieve the PB threshold or failure to collect at least 2.4 × 10⁶ CD34⁺ cells/kg. The primary objective of this single center retrospective study was to evaluate the impact of daratumumab on total HSC yield in transplant eligible NDMM patients. Secondary objectives were the use of plerixafor, the number of apheresis procedures and the number of mobilization failures.
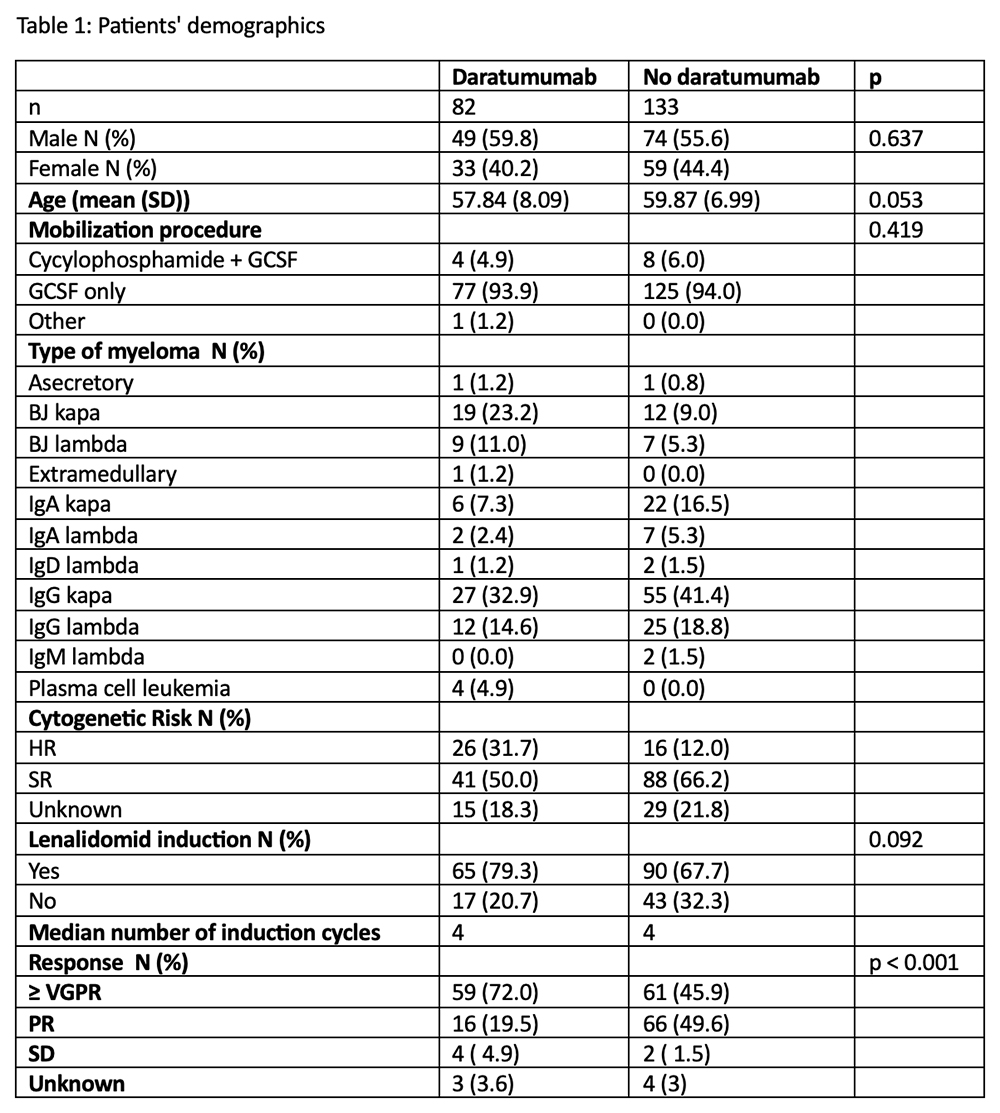
Results. We included 215 patients with NDMM treated at the University Medical Center Ljubljana between January 2022 and February 2025. Patient’s demographics are described in Table 1. Daratumumab‑containing induction regimens were used in 82 of 215 patients (38%). The median PB HSC count on the day of collection was 9.61 × 10⁶/L (IQR 4.7–19.5) in patients receiving daratumumab compared with 20 × 10⁶/L (IQR 12.2–42.9) in those receiving non‑daratumumab induction therapy. Plerixafor rescue was required in 61 of 82 patients (74%) treated with daratumumab, compared with 53 of 133 patients (40%) in the non‑daratumumab group. Administration of plerixafor resulted in a significant increase in PB HSC counts on the following day, with a median of 37.4 × 10⁶ CD34⁺ cells/L (p < 0.001). The median number of apheresis procedures was two, irrespective of daratumumab induction or plerixafor use. The total number of collected HSCs was lower in patients receiving daratumumab‑based induction (5.82 × 10⁶ CD34⁺ cells/kg) compared with those receiving non‑daratumumab induction (6.88 × 10⁶ CD34⁺ cells/kg; p = 0.017). Mobilization failure occurred in five patients (6%) in the daratumumab group and in five patients (4%) in the non‑daratumumab group.
Conclusions. In line with previously published data, we confirmed that patients receiving daratumumab‑containing induction regimens had lower PB HSC counts on the day of collection, resulting in a statistically significantly higher need for plerixafor rescue. With early, on-demand use of plerixafor, the number of apheresis procedures was comparable between groups, with similar rates of mobilization failure. However, patients treated with daratumumab achieved lower HSC yields.

Footnotes
Disclosures
No Conflict of interest.
Funding
No funding.
Article Information

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
