Abstract
Light chain deposition disease (LCDD) is a rare hematologic disorder characterized by the deposition of non-amyloid monoclonal light chains in several organs, with renal involvement representing the main cause of morbidity. Due to its rarity, randomized clinical trials are lacking and no universally accepted standard of care has been established. This review provides a systematic overview of the histological and clinical features of LCDD and summarizes the available therapeutic strategies. In patients with LCDD associated with multiple myeloma, myeloma-based regimens are recommended. In isolated LCDD, chemotherapy with thalidomide, dexamethasone, bortezomib, and autologous stem cell transplantation (ASCT) are commonly employed. In a monocentric experience comparing bortezomib–dexamethasone induction followed by ASCT versus dexamethasone alone, 83% of patients achieved complete hematologic response (CR) at day 100 post-ASCT, with an overall response rate of 100%. At 6 months after ASCT, two-thirds achieved CR, 17% partial response (PR), and 17% no response. All patients achieved a renal response, defined as >50% reduction in proteinuria, and dialysis-free survival at 2 years was 100%. In a UK retrospective cohort of 25 patients, hematologic CR was achieved in 89% of those treated with bortezomib-based regimens, compared with 27% in the thalidomide group. Only one patient treated with lenalidomide achieved PR. Daratumumab has shown promising activity in LCDD. In a small clinical trial including eight patients with concomitant multiple myeloma, 50% achieved very good partial response (VGPR) and 25% achieved renal response. In another series of six patients receiving daratumumab consolidation after VCd induction, one patient improved to hematologic CR, three remained in VGPR, and two remained in PR. Normalization of the free light chain ratio was observed in 50% of patients, compared with none prior to consolidation. Regarding alkylator-based therapy, in a retrospective study of 19 patients treated with melphalan and prednisone, stabilization or improvement of renal function was observed in 5 of 8 patients with baseline creatinine <4.0 mg/dL, whereas 82% of those with creatinine >4.0 mg/dL progressed to end-stage renal disease. A ≥50% reduction in proteinuria was documented in 5 of 15 patients. Although ASCT appears to induce deeper and more durable hematologic and organ responses, no statistically significant superiority over non-transplant approaches has been conclusively demonstrated. Bortezomib-based regimens remain particularly advantageous in patients with renal impairment, as no dose adjustment is required and early hematologic responses are often associated with renal recovery. Renal transplantation is generally discouraged, as disease recurrence occurred in 5 of 7 patients after a median of 33.3 months, with only one patient remaining recurrence-free at 13 years. Overall, LCDD remains a challenging condition requiring individualized therapeutic strategies. Larger prospective multicenter studies with standardized response criteria are needed to define optimal treatment algorithms and long-term outcomes.

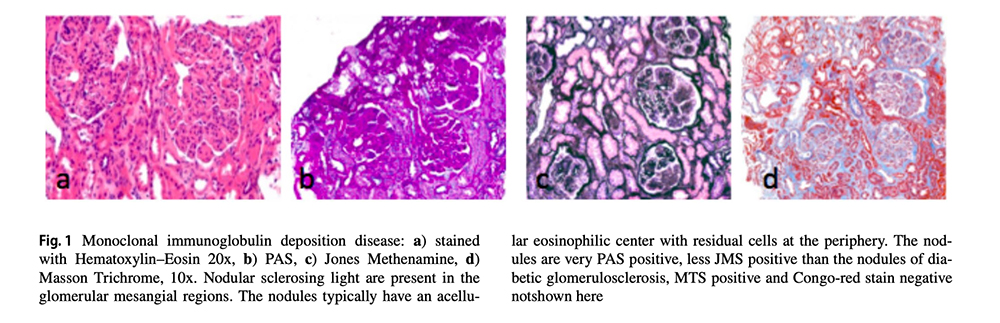
Footnotes
Disclosures
No Conflict of interest.
Funding
No funding.
Article Information

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
