Abstract
Background. The aim of this study was to evaluate safety, tolerability, and clinical activity of belamaf plus lenalidomide and dexamethasone (Rd) (BelaRd) in transplant ineligible newly diagnosed myeloma (TI-NDMM) patients (pts).
Methods. BelaRd trial (NCT04808037) was a phase 1/2, open label study in frail and intermediate-fit pts with NDMM In part 1 (n=36), we evaluated the safety/tolerability of belamaf 2.5/1.9/1.4 mg/kg every 8 weeks (Q8W) plus Rd and we established a recommended phase 2 dose (RP2D) of 1.9 mg/kg Q8W. In Part 2, pts (n=30) were treated at RP2D and randomized 1:1 to Group (Gr) A (dose modification guided by ophthalmologist-assessed KVA scale) or Gr B (hematologist-led Vision-Related Anamnestic (VRA) tool with ophthalmologist assessment focused on grade 3 ≥ ocular adverse events, OAEs).
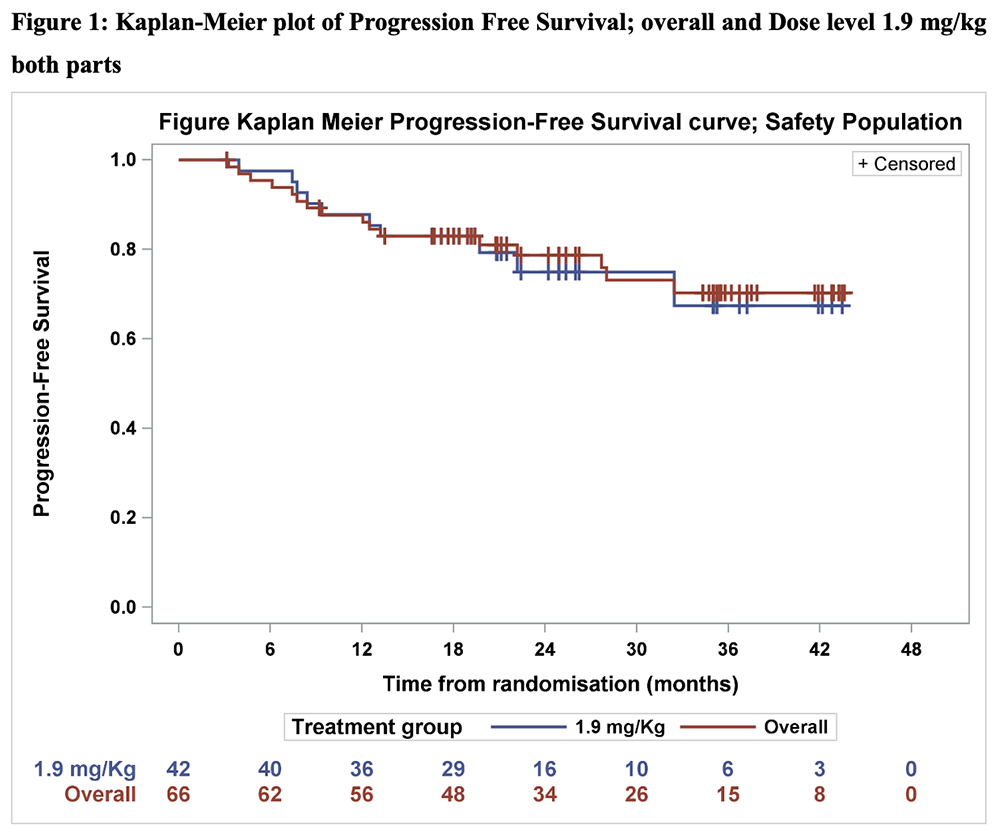
Results. To-date, 42 pts overall (part 1, n=12; part 2, n=30) received belamaf at the RP2D with Rd. Median follow up was 38.4 months (range 13.2-44.1) in part 1 and 19.7 months (range 3.9-26.4) in part 2. Median belamaf exposure was 12 doses in part 1, and 7 doses in part 2. Median dose intensity was 0.6 mg/kg/Q4W in both parts, with median relative dose intensity of 59.4% (range 44.0–89.7) and 51.3% (range 34.1–73.5), respectively. For pts treated at 1.9 mg/kg across both study parts, overall response rate (≥PR) was 97.6% (41/42 pts); 15 pts (35.7%) showed a sCR, 6 pts (14.3%) a CR, 16 pts (38.1%) a VGPR, 4 pts (9.5%) a PR. MRD negativity was achieved in 50% pts (6/12) in part 1, 40% pts (12/30) in part 2. The median time to first response was 1.0 month (range 0.9-3.8) and time to best response was 9.7 months (range 1.0-28.3). Median PFS & TTP were not reached. 18- and 24-month PFS were 82.9% (95%CI: 67.5-91.5) and 74.9% (95%CI: 56.3-86.5), respectively. TTP was 97.2% (95%CI: 81.9-99.6) for both 18- and 24 months. Belamaf administration frequently shifted beyond the normal Q8W schedule due to the OAE-guided holds: within the 1.9 mg/kg cohort (n=42) re-infusion intervals varied widely (8-19 weeks: coefficient of variation (CV) >20% in 32/42 pts) but stabilized beyond 12 months (CV<20% in 21/28 pts). Dose holds occurred in 32.0% in part 1 (OAEs) and in 26.5% in Gr A (OAEs) versus 16.9% in Gr B (VRA-guided); no VRA-enabled dosing was subsequently withheld by ophthalmologists for grade ≥3 OAEs. Grade ≥3 OAEs without VRA-reported “substantial time” (>50% of time) were detected in 10/300 (3.3%) ophthalmologist assessments in Gr A and 2/251 (0.8%) in Gr B. Stopping driving or reading due to eyesight issues occurred in 0.0%-0.6% of the monthly assessments in both groups. Eight pts experienced fatal TEAEs, most commonly pneumonia (3 pts; 7.1%). Grade ≥3 BCVA decline and keratopathy were observed in 54 (13.2%) and 1 (0.3%) of assessments in part 1, and 17 (3.4%) and 1 (0.2%) in part 2, respectively. Grade ≥3 ocular symptoms were observed in 7 pts (58.3%) in part 1 and 9 pts (30%) in part 2. No patient had to discontinue treatment.
Conclusions. Belamaf 1.9 mg/kg Q8W with Rd produced rapid, deep responses and durable disease control with a high ORR and high PFS in TI intermediate-fit/frail NDMM patients. Efficacy was maintained with Q8W schedule having minimal impact on vision related daily activities. Overall, hematologist-led VRA strategy reduced dose holds without compromising ocular safety, supporting streamlined monitoring and warranting randomized phase 3 evaluation of Belamaf-Rd. Funding by GSK (NCT04808037); authors solely responsible for content.
Footnotes
Disclosures
No Conflict of interest.
Funding
No funding.
Article Information

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
