Abstract
Background. In very elderly pts with NDMM data on the efficacy of Isatuximab-lenalidomide-dexamethasone (Isa-Rd) induction therapy followed by Isa-R maintenance therapy compared to Rd induction followed by R maintenance are not available so far.
Aim. In this prospective, randomized study, we aimed to evaluate potential benefits of combining Isa with Rd induction and R maintenance therapy with special focus on results after end of induction therapy.
Methods. Pts were randomized to either 8 cycles of Isa-Rd or Rd; Isa 10 mg/kg on d 1, 8, 15, 22 in cycle 1, subsequently on d 1 and 15; R 25 mg, d 1-21 q 28 d, dexamethasone 40 mg once weekly (20 mg in pts aged ≥75 years). Maintenance therapy was administered for 24 cycles; Isa was given at 10 mg/kg q 28 d, and R at 5-10 mg, d 1-21 q 28 d. MRD was evaluated at the end of induction therapy using NGF with a sensitivity of 10-6. Circulating tumor cells (CTC) were assessed at d 1 and 8 of the study. OS estimates were calculated according to Kaplan-Meier, and survival curves were compared using the log-rank test. This trial is registered on ClinicalTrials.gov (NCT04891809).
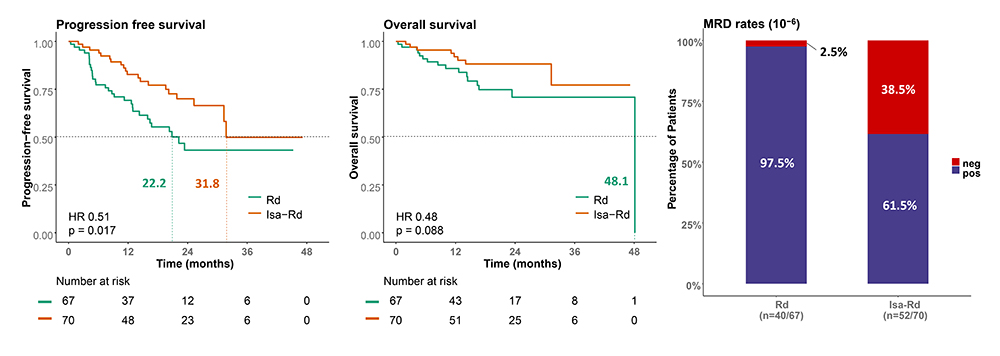
Results. One hundred thirty-seven pts have been enrolled. Median age was 77 yrs (range 70-91 yrs), ISS stage I/II/III: 41 (29.9%)/47 (34.3%)/46 (33.6%), ECOG status 0/1/2: 36 (26.2%)/70 (51.1%)/31 (22.6%). Cytogenetics: t(4;14) 8 (5.8%), t(14;16) 4 (2.9%), del17p 21 (15.3%), ampl1q21 31 (22.6%) of 110-115 pts with results available. Median follow-up was 18.4 mos. The efficacy analysis was performed on the per protocol population, resulting in evaluable response data for 100 pts (50 in each study arm). The median time to best response was 7.3 mos; Isa-Rd (7.0 mos) and the Rd (7.3 mos). All but 8 pts achieved an initial response (≥PR). Response rates were as follows: CR: 26% vs. 10%, p=0.066, VGPR: 36% vs. 20%, p=0.118, PR: 32% vs. 60%, p=0.009, and ORR: 94% vs. 90%, p=0.715, respectively. MR was observed in 6 (6%), and SD in 2 (2%) pts. MRD testing was performed in 92 pts after the end of 8 induction cycles. MRDneg at 10-6 was achieved in 20/52 (34.6%) pts of the Isa-Rd and in 1/40 (2.5%) of the Rd group, resulting in a significant difference in favour of Isa-Rd (p<0.001). MRDneg rates were similar in pts with/without HR cytogenetics. PFS analysis shows a median PFS of 31.8 in the Isa-Rd vs. 22.2 mos in the Rd group, respectively (p=0.017) (Figure 1). A tendency for improved OS was observed in the Isa-Rd group (median OS not reached vs. 48.1 mos, p=0.088; 2-year OS rate 88.2% vs. 70.8%). CTC data were available in 64 pts. Those with CTC detectable at d 8 had significantly shorter PFS (p=0.031). The PFS rate at 24 mos was 28.3% in CTC pos. vs. 73.9% in CTC neg. pts, respectively. Grade ≥3 hematologic AEs, which occurred in ≥10% of pts, were neutropenia (28.5%/19.0%), anemia (24.8%/8.8%), thrombocytopenia (16.8%/5.8%). Grade ≥3 non-haematological AEs in ≥10% of pts, were infections (57.7%/19.7%), pain (43.8%/7.3%), edema (23.4%/0.7%), asthenia (22.6%/4.4%), diarrhea (19.7%/0%), constipation (15.3%/1.5%), rash (14.6%/2.2%), dyspnea (13.9%/1.5%) and polyneuropathy (10.9%/0%). Procedural complications (9 [12.9%] vs. 0, p=0.003), and weight loss (6 [8.6%] vs. 0, p=0.028) were more frequent in the Isa-Rd group.
Conclusions. Our data show a significantly higher CR/VGPR and MRDneg rate and longer PFS and with Isa-Rd compared to Rd (35.4% vs. 2.5%) induction therapy in very elderly pts with NDMM. Updated results will be presented.

Footnotes
Disclosures
No Conflict of interest.
Funding
No funding.
Article Information

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
