Abstract
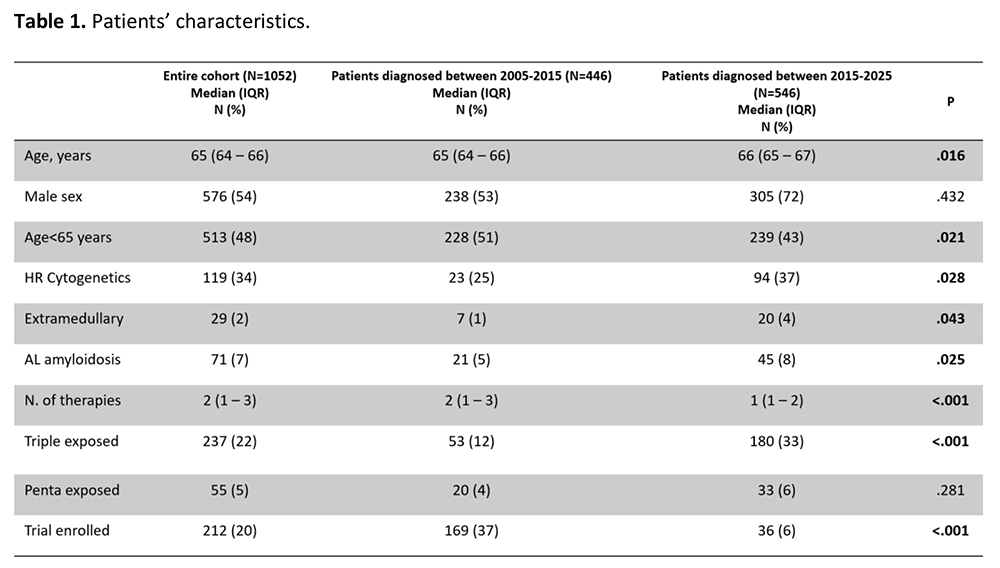
In the last two decades therapeutic armamentarium for treatment of multiple myeloma (MM) is increased both with the introduction of novel drugs and combinations and by introducing consolidation and maintenance therapy. Life expectancy and outcomes of MM patients has remarkably improved. Particularly, maintenance therapy, either such as continuous upfront therapy in transplant ineligible patients or after an induction intensified therapy, has demonstrated to have a pivotal role in improving patient survival either upfront or at relapse. Moreover, the efficacy of new therapies induced a decrease in relapse and refractory (RR) patients We investigated patient characteristics and treatment durations in our center between January 2005 and September 2025. A total of 1052 patients were evaluated. The majority of patients were managed in a real-world setting, outside the context of controlled clinical trials. Patients were divided into two cohorts according to the year of diagnosis, reflecting distinct therapeutic eras. Group A: patients diagnosed and treated between 2005 and 2015; group B: patients diagnosed and treated between 2016 and 2025. Overall survival (OS) was calculated from diagnosis to death or last contact. Time to next treatment (TTNT) was defined as the time from initiating treatment to initiating the next line of therapy. A total of 1052 patients were included: 446 in group A and 546 in group B. Baseline characteristics are reported in Table 1. Median age at diagnosis was 65 years, with 52% of patients aged >65; 54% were male. Cytogenetic data by FISH were available for 497 patients, with greater availability in the later era. Patients received a median of two lines of therapy (range 1–3); approximately 25% were triple-exposed and 5% penta-exposed. Overall, 22% of patients were enrolled in clinical trials. After a median follow-up of 107 months (95% CI: 98–243), median OS for the entire cohort was 65 months (95% CI: 58–69). Considering patients <65 years, median OS was superior in group B compared to group A (not reached vs 75 months, p<0.001). Autologous stem cell transplantation (ASCT) was performed in 338 patients, 43% of whom received tandem ASCT. Patients who underwent ASCT showed a significantly longer overall survival compared with those who did not receive ASCT in both groups (group A: 95 vs 49 months, p<0.001; group B: not reached vs 41 months, p<0.001).Clinical trial participation conferred an OS benefit across both cohorts (group: A 50 vs 80 months, p<0.001; group: B 63 vs 107 months, p <0.001). In RRMM, TTNT progressively shortened with advancing lines of therapy, reaching a median of 4 months after third-line treatment. Beyond the eighth line, TTNT appeared to stabilize, likely reflecting the impact of novel drugs. Sixteen patients received bispecific antibodies across multiple lines (second to tenth); in this subgroup, median progression-free survival was not reached. In the ninth line, TTNT increased from 4 months in the overall population to 11 months in bispecific-treated patients, suggesting a meaningful clinical benefit despite small sample size and limited statistical power. This 30-year single-center real-world analysis highlights the continuous improvement in MM outcomes driven by therapeutic innovation. The sequential integration of IMiDs, proteasome inhibitors, monoclonal antibodies, cellular therapies, and bispecific antibodies has reshaped treatment strategies and survival expectations in routine clinical practice.
Footnotes
Disclosures
No Conflict of interest.
Funding
No funding.
Article Information

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
