Abstract
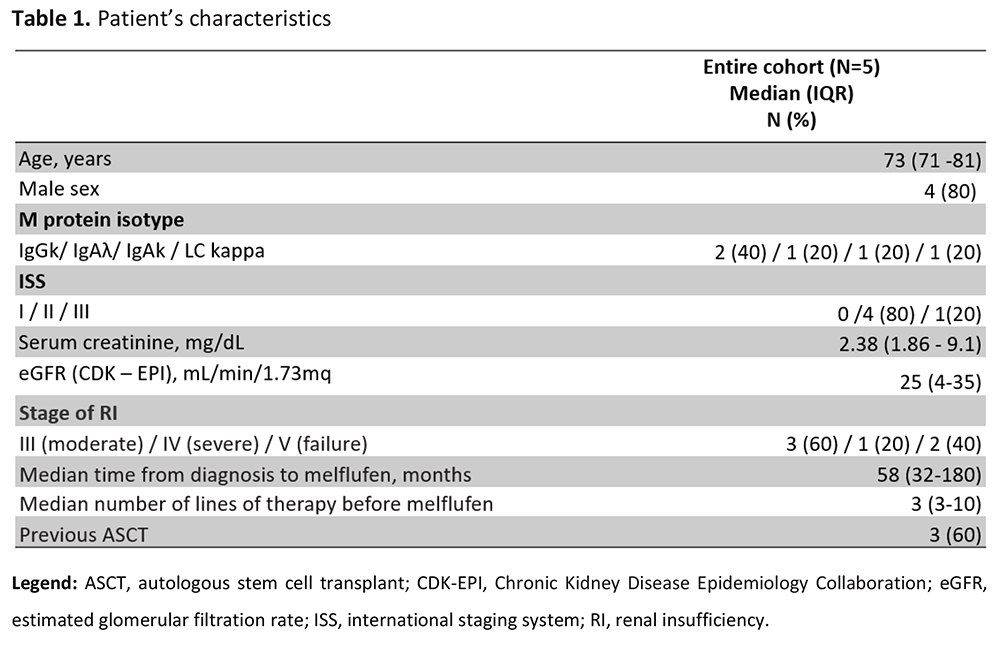
Multiple myeloma (MM) is often complicated with renal insufficiency (RI) which adversely influences the prognosis. The incidence of RI in MM patients ranges from 20% to 50% and is primarily due to light chains deposition in the kidney. Melphalan flufenamide (melflufen) is an anti-myeloma drug approved in Europe for RRMM patients with ≥3 prior lines of therapy. Its efficacy has been demonstrated in phase 2 HORIZON and phase 3 OCEAN trials. Serum creatinine (sCr) above the upper normal limit of 2 mg/dL or an estimated glomerular filtration rate (eGFR) lower than 60 mL/min/1.73 m² defined renal impairment. No dose adjustment of melflufen is required in patients with an eGFR >45 mL/min/1.73 m². A reduced dose of 30 mg is recommended for patients with an eGFR between 30 and 45 mL/min/1.73 m², while insufficient data on safety are available to provide dosing recommendations for patients with an eGFR <30 mL/min/1.73 m². In this study, we retrospectively and prospectively evaluated five relapsed/refractory MM patients with severe renal impairment secondary to multiple myeloma treated with melflufen in our Center. Three of them were primarily treated with high cut-off hemodialysis (HCOD). At baseline, the median concentration of involved serum free light chains (sFLC) was 2340 mg/L, the median sCr level was 2.38 mg/dL, and the median eGFR, calculated using the Chronic Kidney Disease Epidemiology Collaboration (CKD-EPI) creatinine equation, was 25 mL/min/1.73 m². The median time to best hematologic response was 3 months (range: 1–5 months). This was associated with a remarkable reduction in involved sFLC, with a median decrease of 465 mg/L. Alongside, renal function showed significant improvement, with sCr decreasing to a median value of 1.38 mg/dL and eGFR increasing to 48 mL/min/1.73 m². The overall hematological response rate (ORR) was 40%, with 2 patients achieving PR, 2 patients MR and one a stable disease. The overall renal response rate (ORRrenal) was 60%, with 2 patients achieving CRrenal and one patient achieving PRrenal. Two patients (40%) became dialysis independent within a median of 20 days. No severe drug-related toxicities were observed, except for one patient who experienced grade 3 anemia that subsequently resolved. Overall, melflufen was generally well tolerated, demonstrating a favorable safety profile in this subset of patients. This study demonstrates that meflufen could be an effective strategy in rescuing renal function and improving outcomes in patients with RRMM and RI.
Footnotes
Disclosures
No Conflict of interest.
Funding
No funding.
Article Information

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
