Abstract
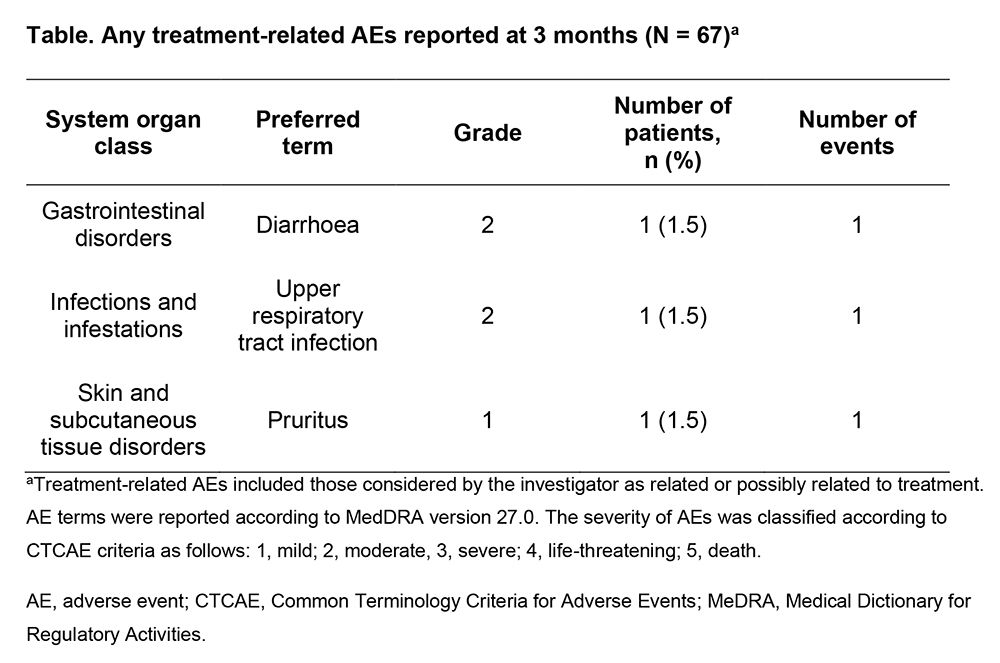
Patients with multiple myeloma (MM) are susceptible to secondary immunodeficiency (SID) owing to both their underlying disease and/or immunosuppressive therapy. HyMMy (NCT05879757) is an ongoing, prospective, non-interventional, observational study of hyaluronidase-facilitated subcutaneous immunoglobulin (fSCIG) 10% in adult patients with MM and SID across 25 centres in Europe. This interim analysis evaluated fSCIG 10% use and associated outcomes at 3-month follow up. Enrolled patients had a diagnosis of MM and met criteria for SID, defined as severe, recurrent or persistent infection(s) despite appropriate anti-infective treatment, and proven specific antibody failure or a serum IgG trough level of < 4 g/L (excluding paraprotein). The primary objective was to assess real-world infusion parameters of fSCIG 10%. Secondary objectives included healthcare resource utilization, MM disease status and adverse events (AEs). Statistical analysis was descriptive. As of 5 May 2025, 67 patients were enrolled; of these, 43 had completed 3 months of follow up. The mean (standard deviation [SD]) age was 64.9 (11.26) years and 53.7% were female. At baseline (enrolment), patients with MM had bone lesions (82.4%), anaemia (49.0%), renal insufficiency (27.5%) or hypercalcemia (13.7%). At baseline, patients received steroids (61.2%), anti-plasma cell monoclonal antibodies (49.3%), immunomodulatory drugs (46.3%), proteasome inhibitors (28.4%) and bispecific antibodies (14.9%). The median (interquartile range [IQR]) time from MM to SID diagnosis was 30 (8.0–67.0) months and the time from SID diagnosis to fSCIG 10% initiation was 2 (0.0–6.0) months. At baseline, 41.6% (57/137) of fSCIG 10% infusions were received every 4 weeks; this increased to 82.2% (97/118) of infusions at 3 months. The mean (SD) dose was 0.36 (0.225) g/kg at baseline and 0.37 (0.319) g/kg at 3 months. At baseline and at 3 months, median (IQR) total infusion volume was 200 (100–250) mL and 300 (210–400) mL, respectively; infusion rate was 133.3 (100–250) mL/h and 210.5 (165–224) mL/h; maximum infusion rate was 240 (240–250) mL/h and 300 (245–300) mL/h; infusion duration was 80 (60–120) minutes and 95 (80–110) minutes; and number of infusion sites was 2 (1–3) and 2.5 (2–3). The shorter infusion duration and fewer infusion sites at baseline were related potentially to dose ramp-up. At baseline and at 3 months, 91.1% (123/135) and 92.2% (129/119) of infusions were in the abdomen, respectively. At baseline, 90.5% (124/137) of infusions occurred in the hospital and 8.8% (12/137) at home; at 3 months, 66.1% (78/118) occurred in hospital and 33.9% (40/118) at home. No infusions were discontinued, slowed or interrupted. Four patients discontinued treatment owing to lack of effectiveness (n = 1) or because they enrolled in another study (n = 3). At 3 months, all treatment-related AEs were mild (1.5%) to moderate (3.0%) in severity, with no serious treatment-related AEs reported (Table). One patient died (2.2%) from an infection unrelated to treatment. This interim analysis shows the feasibility of using fSCIG 10% as a treatment option for patients with MM and SID in real-world setting, potentially offering the flexibility of home infusions with a favourable safety profile. Final study data from up to 12 months of follow-up are needed to confirm these findings.
Takeda Development Center Americas, Inc. funded this study. Takeda Pharmaceuticals International AG funded writing support.

Footnotes
Disclosures
No Conflict of interest.
Funding
No funding.
Article Information

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
