Abstract
Background. Daratumumab-bortezomib-melphalan-prednisone (DVMP) and daratumumab-lenalidomide-dexamethasone (DRd) are standard treatments for transplant-ineligible (NTE) newly diagnosed multiple myeloma (NDMM) patients (pts). No prospective randomized trial has directly compared DVMP vs. DRd. Moreover, real-life older NTE pts are underrepresented in clinical trials.
Aims. We conducted a randomized multicenter phase IV trial (NCT03829371; funded by the Italian Medicines Agency AIFA - Independent Research) to compare safety and efficacy of VMP +/- daratumumab (DVMP) vs Rd +/- daratumumab (DRd) in an unselected real-life population of NTE NDMM pts.
Methods. In the first part of the trial, NDMM pts who were NTE due to age ≥65 years or comorbidities were randomized 1:1 to 9 VMP cycles vs continuous Rd (standard approved schedule). As of July 2022, the protocol was amended to randomize 1:1 pts to DVMP vs DRd. Pts were enrolled regardless of performance status, comorbidities, renal function or baseline laboratory values. Stratification was based on IMWG frailty score and cytogenetic risk [high risk: del(17p), t(14;16) or t(4;14)]. The primary endpoint was progression-free survival (PFS) in the intention-to-treat (ITT) population. Key secondary endpoints included overall survival (OS). Centralized measurable residual disease by next-generation flow (NGF-MRD) was performed in daratumumab-treated pts. In this analysis we focus on data of the daratumumab-treated cohort.
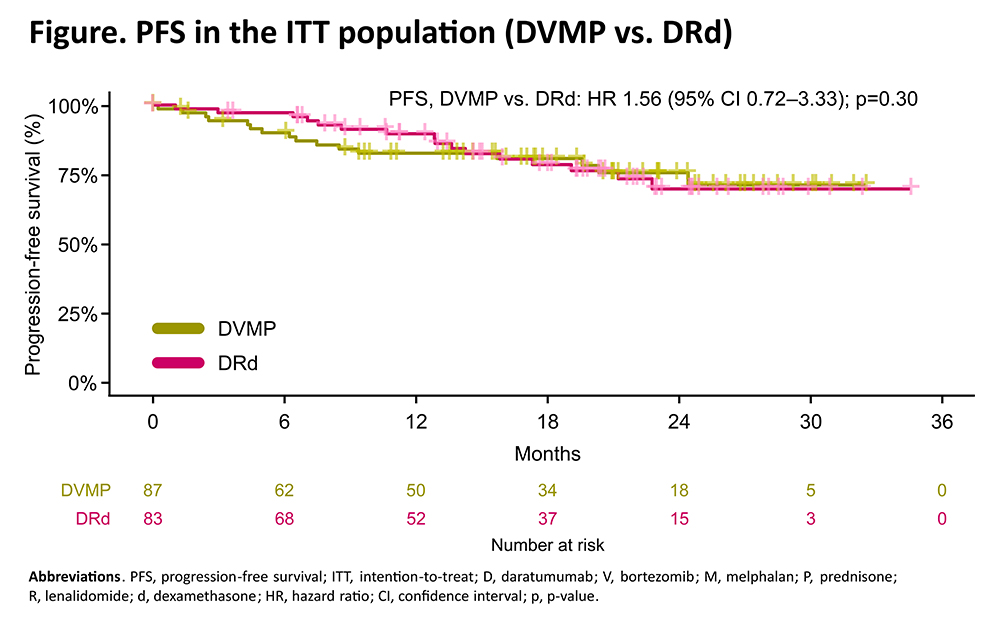
Results. At data cut-off (July 9, 2025), 170 pts received DVMP (n=87) or DRd (83). Baseline characteristics were balanced between DVMP and DRd arms: median age was 76 (range 64–90) vs 76 years (range 63–87); 18% vs 13% of pts were aged >80 years; 37% vs 34% were frail; 27% vs 32% had high-risk cytogenetics. At a median follow-up of 19.3 months, no significant PFS differences were observed between DVMP vs DRd in the ITT population (HR 1.56, 95% CI 0.72–3.33, p=0.30; Figure). No significant differences were observed across age (> or ≤80 years), IMWG frailty score or cytogenetic-defined risk subgroups. The 6-month and 1-year PFS rates were 90% vs 97% and 83% vs 90% with DVMP vs DRd. In the first 6 months, 9 PFS events (2 progressive disease and 7 deaths) were observed [7/9 (78%) pts were frail; 7/9 (78%) events were observed in the DVMP arm]. In the ITT population, the 12-month NGF-MRD negativity rate was 25% with DVMP vs 30% with DRd (OR 1.79, 95% CI 0.81–3.94, p=0.15). Reaching MRD negativity within 12 months led to an improved PFS (vs MRD positivity: HR 0.12, 95% CI 0.03–0.50, p=0.004). No new safety concerns were reported.
Conclusion. We confirmed the efficacy of DVMP and DRd in an older real-life NTE NDMM population including ~35% of frail pts. At current follow-up, no significant PFS differences were observed between DVMP and DRd, but early mortality was more frequent in frail and DVMP-treated pts. Centralized MRD assessment in this real-life setting was feasible, and MRD negativity rates were comparable to those in registrational trials.

Footnotes
Disclosures
No Conflict of interest.
Funding
No funding.
Article Information

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
