Abstract
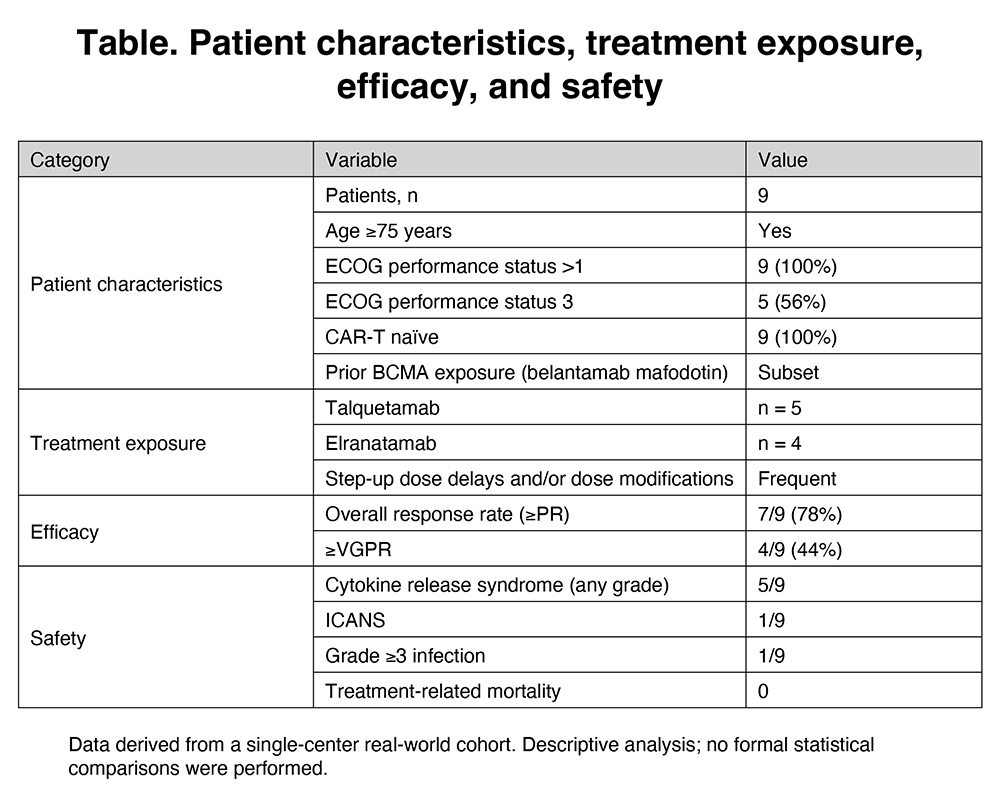
Bispecific antibodies (BsAbs) have demonstrated high efficacy in relapsed/refractory multiple myeloma (MM). However, patients with impaired performance status and clinical frailty are underrepresented in pivotal trials, which typically restrict enrollment to ECOG 0–1. As a result, evidence guiding BsAb use in frail and elderly patients – who represent a substantial proportion of real-world MM populations – remains limited. In particular, the feasibility of BsAb therapy in patients with ECOG >1, the need for treatment adaptations, and the impact of supportive care strategies are insufficiently characterized. We conducted a single-center real-world analysis of consecutive frail and elderly MM patients with ECOG performance status (PS) >1 treated with BsAbs between 2024 and 2026. Frailty was defined pragmatically by age ≥75 years and/or ECOG PS ≥2. Patients received talquetamab or elranatamab according to institutional practice. All patients were CAR-T naïve, and a subset had prior BCMA exposure with belantamab mafodotin. Data collection included baseline clinical characteristics, treatment exposure, step-up delays and dose modifications, safety outcomes, and treatment response assessed according to standard clinical criteria. All patients received immunoglobulin replacement therapy. In addition, viral (including CMV) and fungal molecular surveillance was performed prior to treatment initiation and at each febrile episode. Analyses were descriptive, with exploratory subgroup evaluation of patients with ECOG PS 3. Nine patients were included; five received talquetamab and four elranatamab. Five patients (56%) belonged to a high-risk ECOG PS 3 subgroup. Step-up dose delays and/or dose modifications were frequently required, reflecting advanced age, comorbidities, and functional impairment. Overall response rate was 78% (7/9), with ≥VGPR achieved in 44% (4/9). Responses were observed across the ECOG >1 population, including within the high-risk ECOG PS 3 subgroup, despite frequent treatment adaptations. Treatment initiation was feasible in all patients, including those with advanced frailty and ECOG PS 3. No patients discontinued therapy during step-up dosing due to intolerance, and treatment exposure was maintained despite frequent dose adaptations. Dose delays and modifications were implemented primarily to mitigate toxicity risk rather than in response to severe adverse events. Cytokine release syndrome occurred in five patients and was predominantly low-grade. Immune effector cell-associated neurotoxicity syndrome was observed in one patient. One patient experienced a grade ≥3 infection. No unexpected toxicities, early treatment discontinuations due to toxicity, or treatment-related mortality were observed. In this real-world cohort of frail and elderly MM patients with ECOG >1, BsAb therapy demonstrated clinically meaningful efficacy with manageable toxicity when individualized dosing and proactive supportive care strategies were applied. Poor performance status alone should not automatically preclude BsAb therapy. These findings support the feasibility of BsAbs in frail populations traditionally excluded from clinical trials and highlight the need for prospective studies specifically addressing this high-risk group.
Footnotes
Disclosures
No Conflict of interest.
Funding
No funding.
Article Information

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
