Abstract
Introduction. Teclistamab is the first approved BCMA×CD3 bispecific antibody for treating triple class exposed (TCE) relapsed/refractory multiple myeloma (RRMM) with ≥20,800 patients treated worldwide. LocoMMotion and MoMMent are prospective, multinational, non interventional studies of real world physician’s choice (RWPC) in TCE RRMM, specifically designed to serve as external control arms for comparison with novel therapies. A prior comparative analysis of MajesTEC-1 versus pooled LocoMMotion + MoMMent data showed significantly improved efficacy with teclistamab compared with RWPC. To confirm its relative effectiveness in routine clinical practice, we compared data from REALiTEC, an international retrospective observational study of teclistamab use outside of clinical trials, with pooled data from LocoMMotion + MoMMent.
Methods. Using individual patient data from both cohorts, the effectiveness of teclistamab in REALiTEC versus RWPC was determined. Inverse probability of treatment weighting (IPTW), using the average treatment effect (ATE) approach, was implemented to adjust for imbalances in 15 baseline covariates of prognostic significance. The relative effectiveness of teclistamab versus RWPC for response rates was estimated with an odds ratio using weighted logistic regression transformed into a response-rate ratio (RR) and 95% confidence interval (CI). Weighted proportional hazards regression was used to estimate hazard ratios (HRs) and 95% CIs for duration of response (DOR), progression-free survival (PFS) and overall survival (OS). Multivariable logistic and proportional hazards regression including covariates were done as sensitivity analyses. For all studies, patients provided written informed consent, and an independent ethics committee or institutional review board at each study center approved the study protocol.
Results. 113 heavily pre-treated patients from REALiTEC, mainly treated via pre-approval access programs (88.5%), and 340 patients from LocoMMotion and MoMMent studies were included in the analysis. Median follow-ups were 20.7 months and 24.3 months, respectively. Before reweighting, differences were observed in several baseline characteristics, with REALiTEC having a higher proportion of patients with prior BCMA exposure (35.4% vs 16.8%), triple-class refractory (34.5% vs 20.6%), >4 prior lines (69.9% vs 49.4%), and having a prior transplant (78.8% vs 65.9%). Conversely, REALiTEC had fewer renally impaired patients (creatinine clearance ˂60mL/min: 26.5% vs 39.7%). After reweighting, baseline characteristics were well balanced between cohorts. Patients treated with teclistamab had significantly higher and deeper response rates as compared to those treated with RWPC: ORR (RR [95% CI], 2.06 [1.64–2.58]; p<0.0001), ≥VGPR (RR [95% CI], 4.03 [2.85–5.70]; p<0.0001), ≥CR (RR [95% CI], 38.69 [8.57–174.72]; p<0.0001). Similarly, IPTW-adjusted DOR (median 26.1 months vs 11.1 months; HR: 0.53 [0.32-0.87], p=0.0123), PFS (median 10.58 months vs 4.17 months; HR 0.45 [0.33–0.63], p˂0.0001), and OS (median NE vs 14.42 months; HR 0.59 [0.41–0.85], p=0.0043) were significantly longer with teclistamab versus RWPC (Figure 1). Results from the multivariable analyses were consistent.
Conclusions. Teclistamab demonstrated significantly improved effectiveness in REALiTEC over RWPC in LocoMMotion + MoMMent, emphasizing its clinical benefit as a highly effective treatment for patients with TCE RRMM in routine clinical practice. 
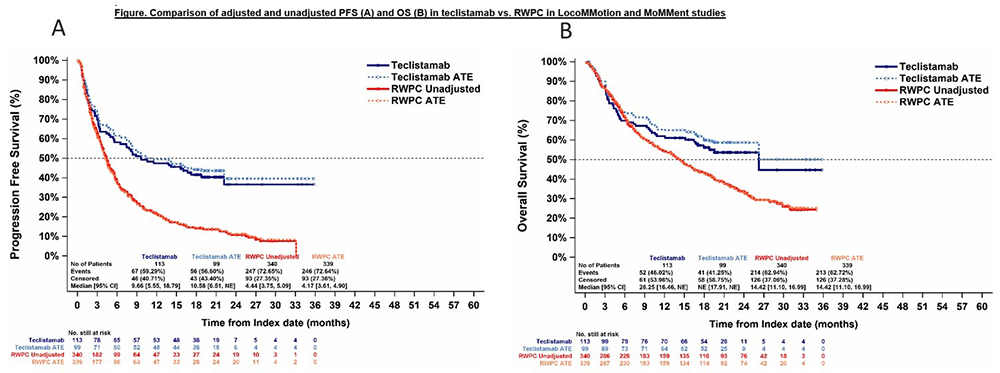
Footnotes
Disclosures
No Conflict of interest.
Funding
No funding.
Article Information

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
