Abstract
Background. Depth of response is a central determinant of clinical outcomes in multiple myeloma (MM), and bone-marrow-based minimal residual disease (MRD) has emerged as one of the strongest surrogate biomarkers for progression-free (PFS) and overall survival (OS). However, MRD assessment remains invasive, potentially spatially biased, resource-intensive and logistically challenging, restricting its practicability for frequent longitudinal disease monitoring and large-scale implementation in routine practice. Mass spectrometry (MS)-based detection of monoclonal proteins in peripheral blood represents an emerging, promising and minimally invasive alternative. Matrix-assisted laser-desorption/ionization time-of-flight (MALDI-TOF) MS, a top-down light-chain-based method, enables highly sensitive identification and tracking of patient-specific M-proteins, discrimination from therapeutic antibodies, and accurate quantification below the detection limits of conventional serum protein electrophoresis (SPEP) and immunofixation (IFE). While earlier studies (e.g., Puig et al. 2024; Kubicki et al. 2024; Mai et al. 2023; Claveau et al. 2022) already suggested a clinically significant prognostic role for MS-based measurable residual activity (MRA), large prospective evaluations within randomized phase 3 trials are currently limited. The present study addresses this critical gap by evaluating MS-based MRA longitudinally in a large, well-characterized cohort of patients treated within the phase 3 GMMG-HD7 trial.
Methods. Serum MS was performed on 3,301 samples from 617 patients enrolled in the phase 3 GMMG-HD7 trial at baseline, post-induction (IND), post-autologous stem cell transplant (ASCT), 12 and 24 months of maintenance (12M MT, 24M MT), end-of-study (EOS), and at progressive disease (PD). MS-based MRA was assessed using the EXENT® system. Bone-marrow MRD was evaluated via Next-Generation Flow cytometry (sensitivity 10-5). Prognostic impact on progression-free survival (PFS) was analyzed at predefined timepoints, alone and in combination with MRD. Multivariate Cox models were fitted considering age, sex, first randomization treatment arm and risk scoring (R-ISS), separately for every landmark.
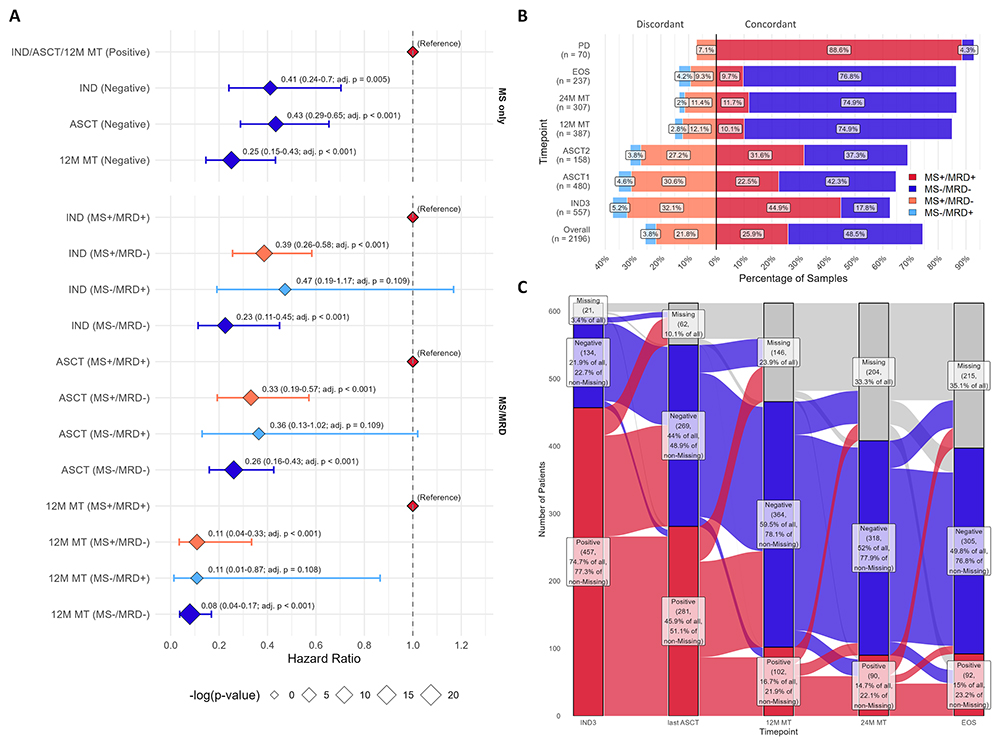
Results. As seen in Figure 1, MS reliably distinguished therapeutic antibodies from patient M-proteins and accurately tracked longitudinal M-protein dynamics. MS negativity strongly predicted superior PFS, most prominently during maintenance (12M MT: HR 0.25; 95% CI 0.15–0.43; adjusted p<0.001). Combined MS/MRD assessment further refined risk stratification: double-positive patients had the poorest outcomes, whereas all other groups exhibited comparably favorable PFS. Concordance between MS and MRD increased over time (post-IND: 62.7%; 12M MT: ~85%), with early discordance influenced by IgG recycling. MS detected low-level residual M-protein with higher sensitivity than serum protein electrophoresis (SPEP), enabling quantification in samples at sub-gram ranges below conventional assay limits. MS/MRD/SPEP/IFE concordant samples were most frequent with all-negative profiles predicting the best outcomes.
Conclusions. Serum MS is a sensitive, reproducible, and practical biomarker for detecting and tracking residual disease in MM. It complements MRD, improves risk stratification, and allows minimally invasive monitoring. Integration of MS-based MRA into future response criteria and risk-adapted treatment strategies may define deeper remission states and guide therapy decisions in MM.

Footnotes
Disclosures
No Conflict of interest.
Funding
No funding.
Article Information

This work is licensed under a Creative Commons Attribution-NonCommercial 4.0 International License.
